- SFA-002 Updated Extension Data Showing 92% PASI 50 and 72% PASI 75
- $4.5 Million Bridge Financing led by North South Ventures
- Composition of Matter patent which covers over 40 potential disease indications for SFA-002
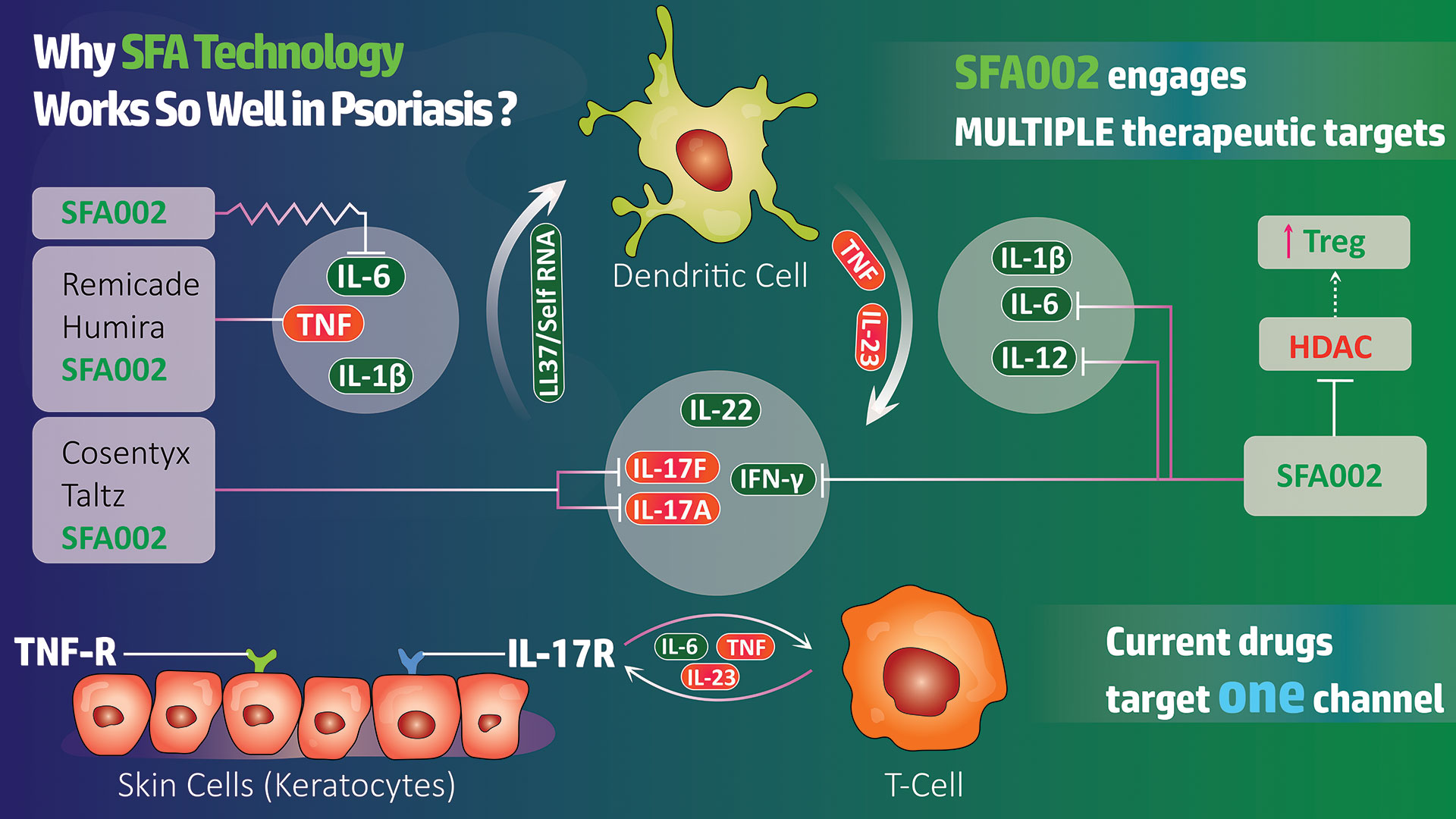
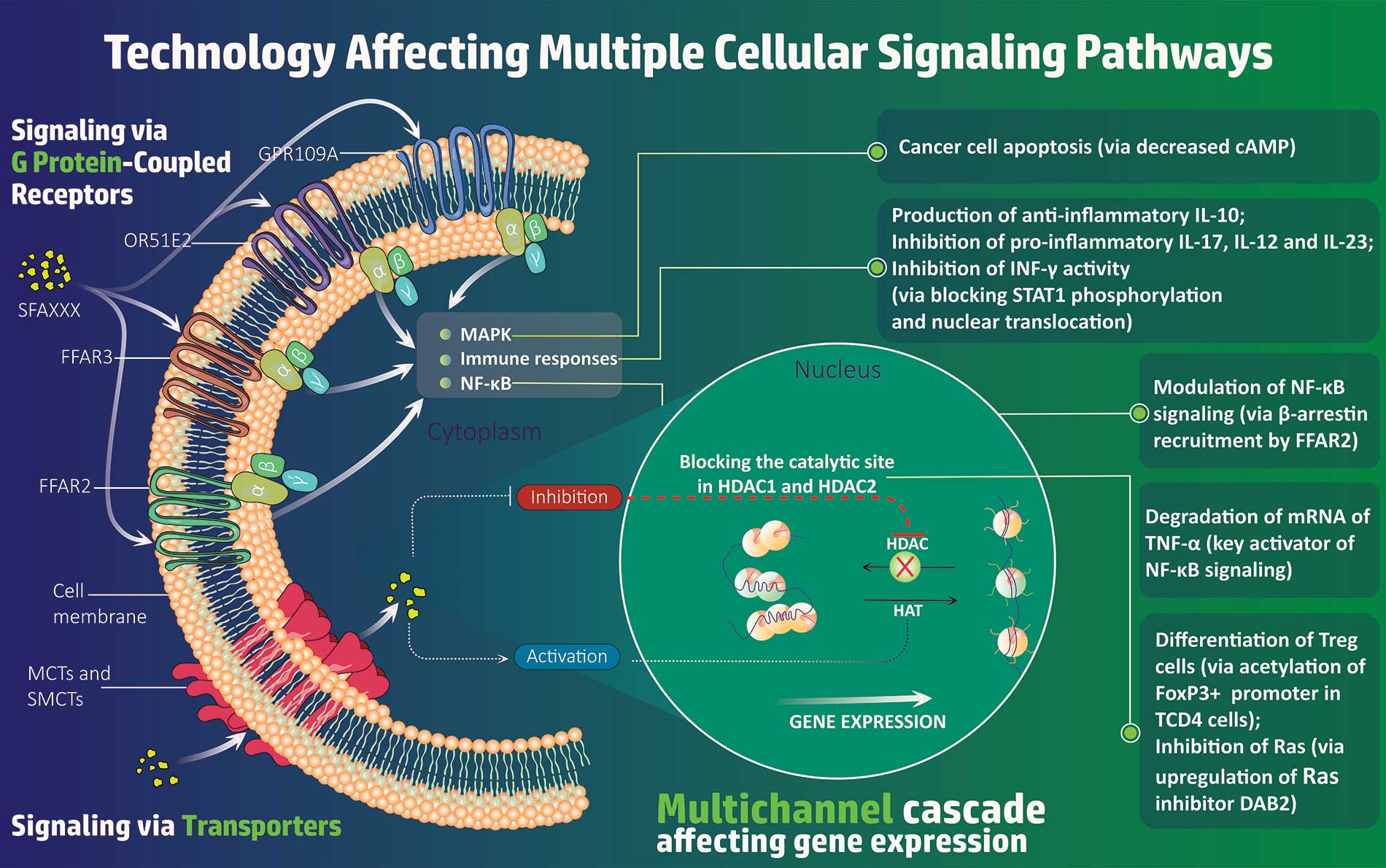
JENKINTOWN, Pa., Nov. 09, 2023 (GLOBE NEWSWIRE) — SFA Therapeutics, a clinical-stage biotech using a first-in-class immuno-modulation technology platform, is pleased to announce the completion of a bridge financing of $4.5 million, led by North South Ventures. Additional participants in this raise were the Ben Franklin Technology Partners of Pennsylvania, Savantus Ventures and Asymmetry Ventures. The proceeds from the financing allow for advancement of lead program SFA-002 through ongoing Phase 1b cohort 2 (15 additional psoriasis patients) with the final data expected in the first half of 2024 and planning for the Phase 2 clinical trial development. SFA-002 is an oral pill that simultaneously acts on multiple therapeutic pathways, unlike other drugs on the market which target a single channel. SFA-002 provides an immunomodulatory effect by down-regulating levels of pro-inflammatory cytokines involved in the pathogenesis of psoriasis including TNF-α, IL-23, IL-10, IL-12, IL-17 and INF-γ, and down-regulating autoimmunity. SFA-002 has the potential to be disease modifying and will create a new class of immunomodulators that work by blocking autoimmunity instead of immunosuppression.
The Phase 1b clinical trial of SFA-002 (ClinicalTrials.gov Identifier: NCT05642182) in mild-to-moderate chronic plaque psoriasis is an open label prospective study of 2 different formulations, in two cohorts. The study protocol is 12 weeks of active therapy, one month of follow-up and an optional 12 weeks of extension. The primary outcome measure is safety, and the secondary efficacy outcomes are the IGA score (International Global Assessment) and Psoriasis Area and Severity Index (PASI) score after study completion.
Today’s updated data from the SFA-002 Phase 1b cohort 1 three-month extension clinical trial are as follows:
SFA-002 Phase 1b Cohort 1 Extension Analysis Highlights:
In 14 evaluable subjects (including 6 subjects on the 3-month extension)
- 92% of patients demonstrated a PASI improvement of greater than 50
- 72% of subjects showed an improvement of greater than PASI 75
- Two subjects achieved PASI 100 (100% clearance)
- Onset of action was observed in as little as 6-weeks
- Clinically meaningful treatment effects included patients with difficult to treat scalp psoriasis and palmoplantar psoriasis
- No related adverse events or toxicities were observed during the treatment period and follow-up, with no rebound effects observed
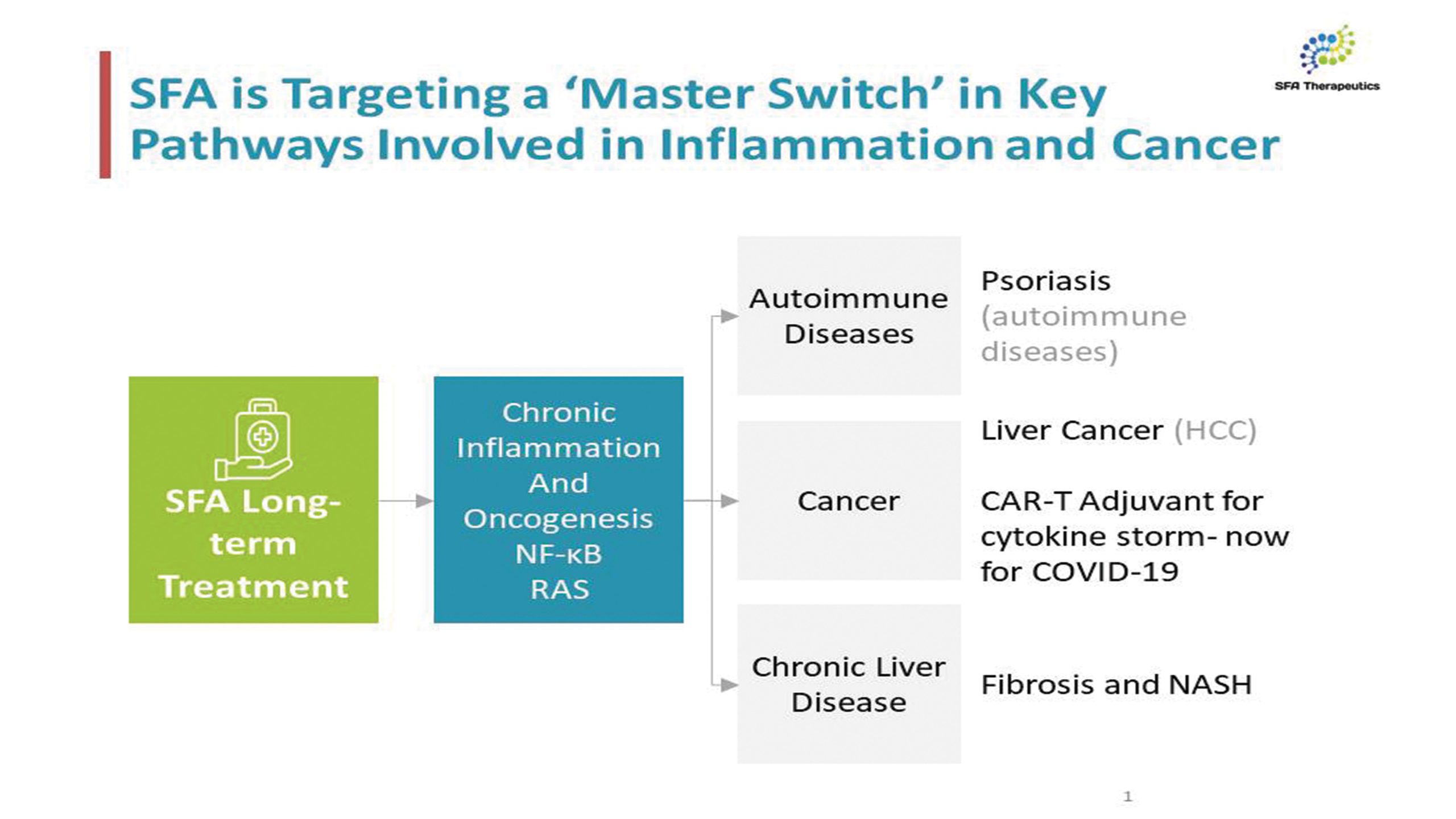
SFA Therapeutics was also recently granted a Composition of Matter patent, US patent number 11,759, 442 B2, issued on September 19, 2023. This foundational patent covers over 40 potential disease indications for SFA-002, a drug which works by blocking autoimmunity, a new approach which has the potential to significantly change the treatment of autoimmune diseases.
“The very encouraging results from this SFA-002 cohort provide further evidence for the platform that SFA Therapeutics is building around novel formulations of short-chain fatty acids, and the latest patent clearly strengthens their position as global leaders in this field,” stated David Naylor, MD/PhD of North South Ventures.
SFA Therapeutics is also pleased to announce that North South Ventures Managing Partner, Dr. David Naylor, MD/PhD, OC, FRCPC, FRSC, has joined SFA Therapeutics’ Board of Directors. Dr. Naylor is an eminent physician/scientist who is the former President of the University of Toronto, and has conducted significant research in a number of important diseases.
Psoriasis is an incurable chronic disease that affects 125 million people worldwide, with the global psoriasis treatment market size projected to grow at a CAGR of 9.2%. The current gold-standard treatment for autoimmune disease is single-pathway, injectable immunosuppression. Due to affordability and accessibility, as many as 90% of patients with autoimmune diseases do not take immunosuppressants. Over 30% of patients on immunosuppression therapy do not respond to treatment. Further, the side effects of immunosuppression include susceptibility to infectious diseases such as TB and rare cancers.
About SFA Therapeutics, Inc.
SFA Therapeutics is a clinical-stage biotech using a first-in-class immuno-modulation platform to discover and develop next generation therapies, focused on the treatment of autoimmune diseases. SFA Therapeutics is advancing a focused portfolio of clinical and pre-clinical assets to optimize efficacy and safety in multiple inflammatory diseases, afflicting over 350 million patients worldwide. SFA-002 is the lead asset in the SFA Therapeutics portfolio, serving as a proof-of-concept for SFA’s key mechanism of action aimed at treating a range of autoimmune diseases.
Contacts:
INVESTORS
Mike Moyer
LifeSci Advisors
mmoyer@lifesciadvisors.com
COMPANY
Ira C. Spector, PhD
CEO
SFA Therapeutics, Inc.
iraspector@sfatherapeutics.com
Source: SFA Therapeutics, Inc.