⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.
Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
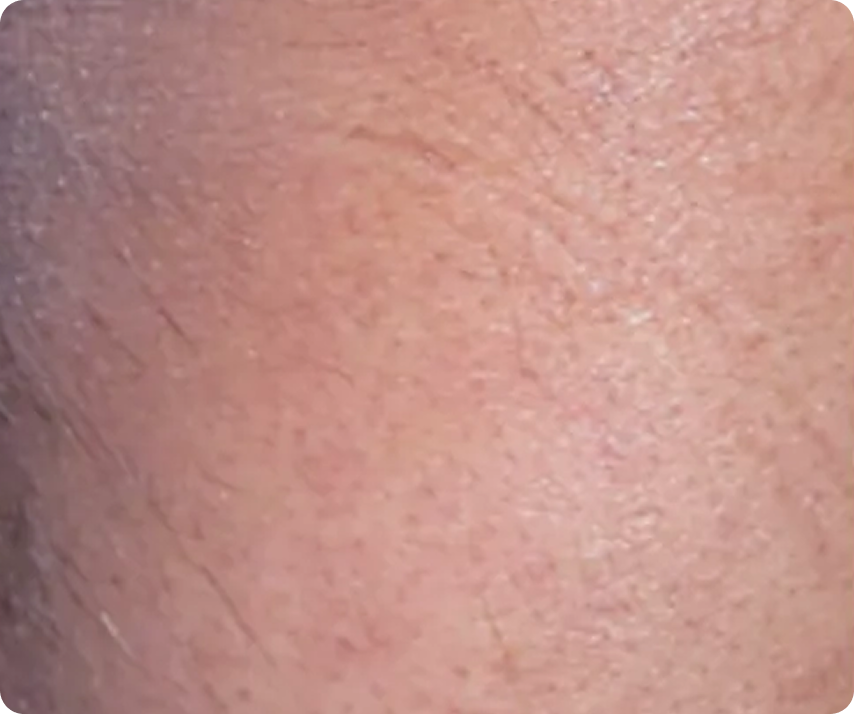
Representative Clinical Photography
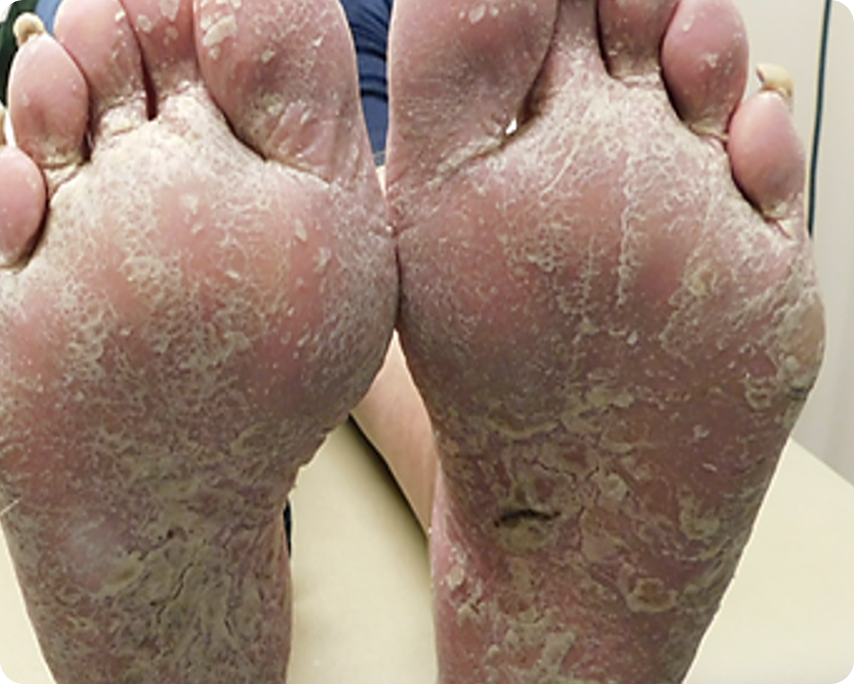
PASI 100
Baseline vs. Week 12
Complete clearance
Before
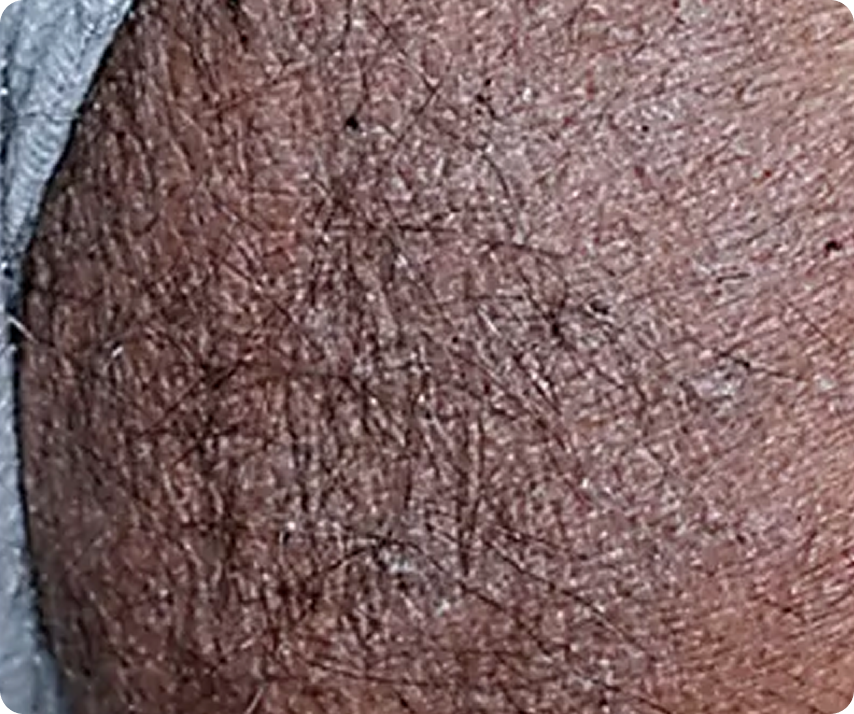
After
PASI 90
Baseline vs. Week 12
High responder
Before
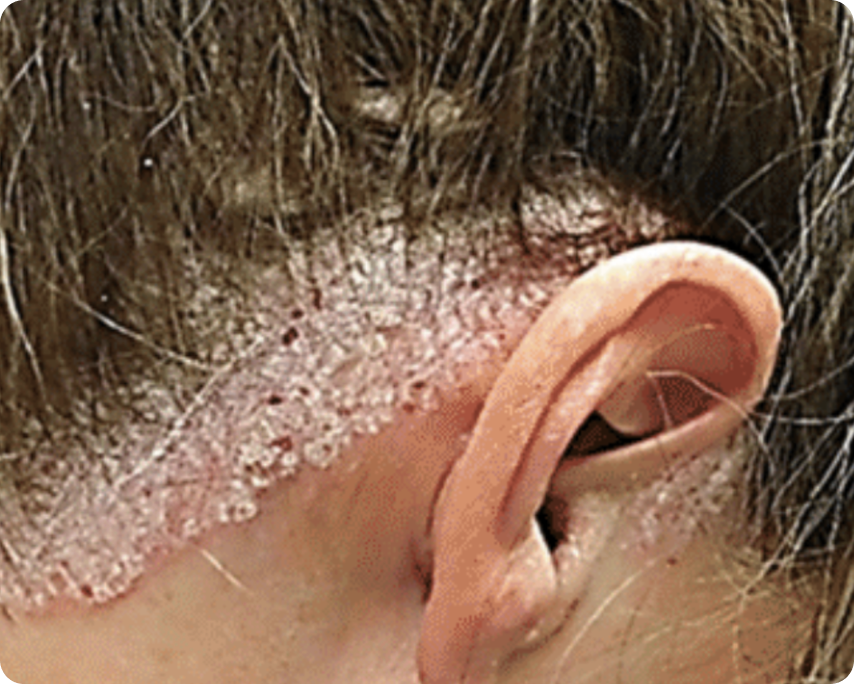
After

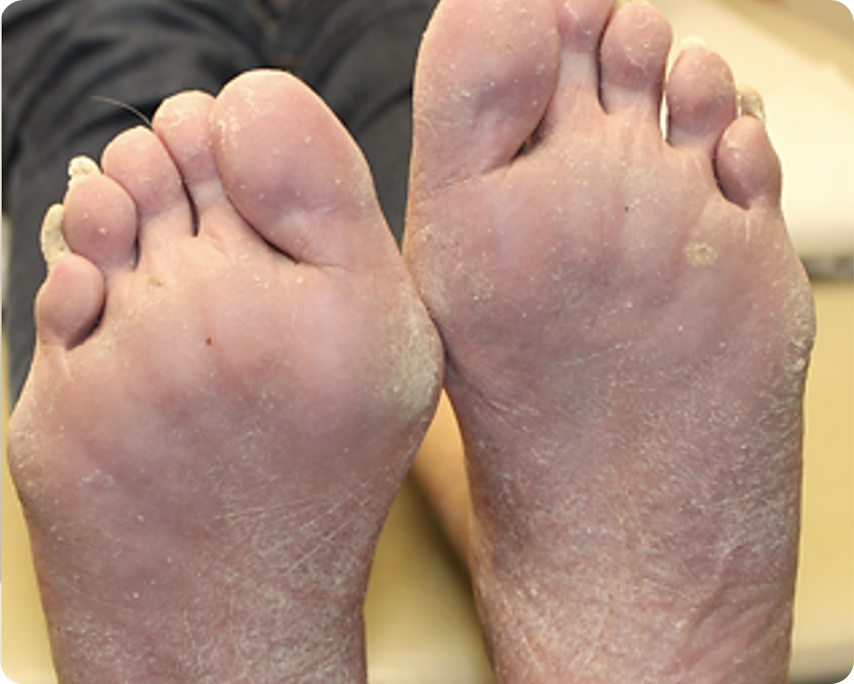
PASI 75
Baseline vs. Week 12
Typical responder
Before
After
Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].
⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.

Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
Representative Clinical Photography
PASI 100
Baseline vs. Week 12
Complete clearance
Before

After

PASI 90
Baseline vs. Week 12
High responder
Before

After

PASI 75
Baseline vs. Week 12
Typical responder
Before

After

Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
Asset
Indication
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].
⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.

Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
Representative Clinical Photography
PASI 100
Baseline vs. Week 12
Complete clearance
Before

After

PASI 90
Baseline vs. Week 12
High responder
Before

After

PASI 75
Baseline vs. Week 12
Typical responder
Before

After

Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
Asset
Indication
Progress
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].
⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.

Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
Representative Clinical Photography
PASI 100
Baseline vs. Week 12
Complete clearance
Before

After

PASI 90
Baseline vs. Week 12
High responder
Before

After

PASI 75
Baseline vs. Week 12
Typical responder
Before

After

Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
Asset
Indication
Progress
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].
⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.

Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
Representative Clinical Photography
PASI 100
Baseline vs. Week 12
Complete clearance
Before

After

PASI 90
Baseline vs. Week 12
High responder
Before

After

PASI 75
Baseline vs. Week 12
Typical responder
Before

After

Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
Asset
Indication
Progress
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].
⚠ SFA-002 is an investigational therapy. It has not been approved by the FDA or any regulatory authority. Safety and efficacy have not been established.

Resetting the Immune System in Psoriasis
SFA-002 is an oral deuterated short-chain fatty acid designed for once-daily dosing that resets immunity through the gut-immune axis. A new chemical entity with a clean safety profile and Phase 1b data showing significant clearance in plaque psoriasis.
Phase 1b
Clinical Stage
28
Participants Enrolled
18
Granted Patents
NCE
New Chemical Entity
Phase 1b Results: SFA-002 in Plaque Psoriasis
Open-label safety study in 28 participants with mild to severe plaque psoriasis over 12 weeks. Results demonstrate significant response rates across all primary endpoints.

Significant Clearance
Statistically significant response rates observed across all primary efficacy endpoints

Clean Safety Profile
No treatment-emergent adverse events reported in any participant
Safety highlight: No treatment-emergent adverse events observed in any of the 28 participants. Endogenous pathway with 10x safety margin below NOAEL.Over 100 human exposures across the SFA platform.
Important: SFA-002 is an investigational therapy. These results are from an open-label Phase 1b study (n=28) and have not been confirmed in a randomized, placebo-controlled trial. Phase 2 study is in preparation. Individual results may vary. SFA-002 has not been approved by the FDA or any regulatory authority.
Representative Clinical Photography
PASI 100
Baseline vs. Week 12
Complete clearance
Before

After

PASI 90
Baseline vs. Week 12
High responder
Before

After

PASI 75
Baseline vs. Week 12
Typical responder
Before

After

Important: Images are from an open-label Phase 1b study (n=28) and are representative of individual participant outcomes, not guaranteed results. Patient consent obtained for all clinical photography. SFA-002 is an investigational therapy and has not been approved by the FDA or any regulatory authority.
How SFA-002 Resets Immunity
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Step 1
Oral administration
Once-daily deuterated SCFA tablet reaches the gut, where 70% of the immune system resides
Step 2
IL-10 Upregulation
SFA-002 raises interleukin-10, the body's key anti-inflammatory cytokine
Step 3
T-reg Expansion
Expanded regulatory T cells suppress the autoreactive immune response driving psoriasis
Step 4
Immune Reset
Resets immunity with sustained reductions in IL-17, IL-23, IFN-γ, and TNF
Pipeline: Deuterated Short-Chain Fatty Acid Therapeutics
Our proprietary deuteration platform creates patentable new chemical entities from naturally occurring short-chain fatty acids, unlocking NCE exclusivity and once-daily dosing.
Asset
Indication
Progress
SFA-002
Oral, once-daily tablet
Plaque Psoriasis (Mild-to-severe)
Phase 2
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-009
Oral
Pancreatic Cancer (combination therapy)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
SFA-001
Oral
Liver Cancer (chronic liver disease to hepatocellular carcinoma)
PRECLINICAL
PHASE 1
PHASE 2
PHASE 3
Future
Oral
Rheumatoid Arthritis, Inflammatory Bowel Disease, Additional Autoimmune
DISCOVERY
PHASE 1
PHASE 2
PHASE 3
Phase 2 Clinical Trial
A randomized, double-blind, placebo-controlled study of SFA-002 in 120 patients with mild to severe plaque psoriasis.
Get study updates
Leadership
Experienced Drug Development Team
Shawn O'Brien
Chairman
8 dermatology deals at Cipher Pharmaceuticals, 9 NDAs. Deep derm commercialization and M&A expertise.
Ira C. Spector
Co-Founder & Chief Scientific Officer
Physicist turned epidemiologist/biostatistician. Helped create Wyeth, transform Allergan (acquired by AbbVie). Ran all Enbrel trials, $18B franchise across 8 indications.
Mark Feitelson
Co-Founder & Scientific Advisor
Named lab at Temple University. Pioneered "What do gut microbes produce, and can we turn those into drugs?" Inventor of the SFA platform.
Alla Arzumanyan
Co-Founder & Chief Development Officer
Drug development and infectious disease research. Inventor of SFA-002.

Resetting the immune system © 2026 SFA Therapeutics, Inc.
SFA-002 and all SFA Therapeutics pipeline candidates are investigational therapies and have not been approved by the U.S. Food and Drug Administration (FDA) or any other regulatory authority. The safety and efficacy of these therapies have not been established. The information on this website is intended for informational purposes only and should not be construed as medical advice or an offer to participate in a clinical trial. ClinicalTrials.gov Identifier: [pending registration].