
Oral immune tolerance therapy for psoriasis
An investigational oral therapy designed to restore immune tolerance in psoriasis, supported by early human results.
See psoriasis results
Phase 1b Study of SFA-002 | 28 Participants | 12 Weeks
Early human results showed meaningful skin clearance in people with mild to severe plaque psoriasis.
Efficiency
Phase 1B results demonstrated statistically significant response rates competitive with approved oral and biological therapies.
Safety
No treatment-emergent adverse events were observed in 28 participants during the phase 1b, suggesting SFA-002 is highly suitable for mild plaque psoriasis patients.
* Phase 1b, open-label study. Safety assessed through treatment-emergent adverse events.
Individual Results
Results from Phase 1b. Individual results vary.
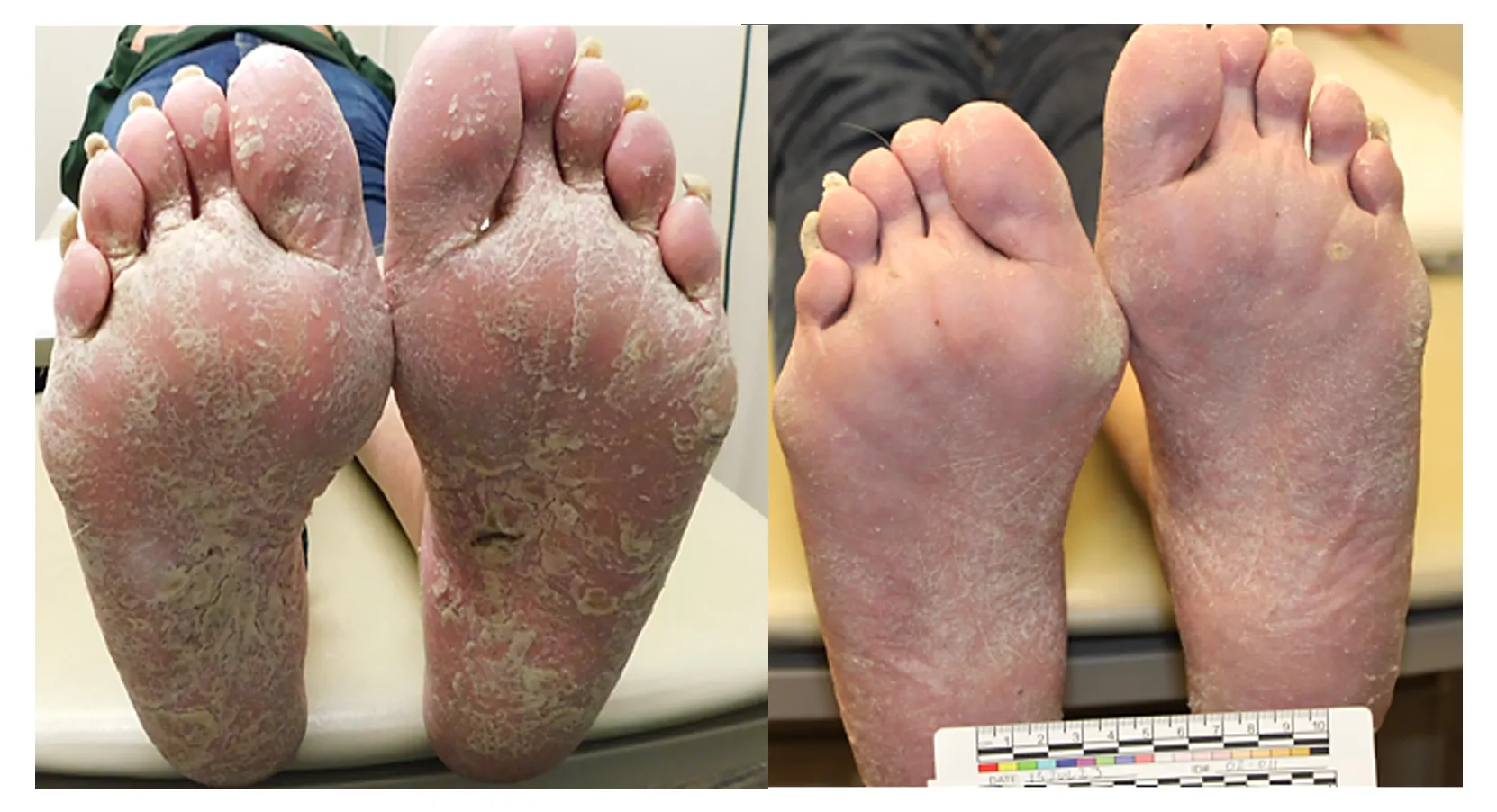
(PASI 100) Complete clearance
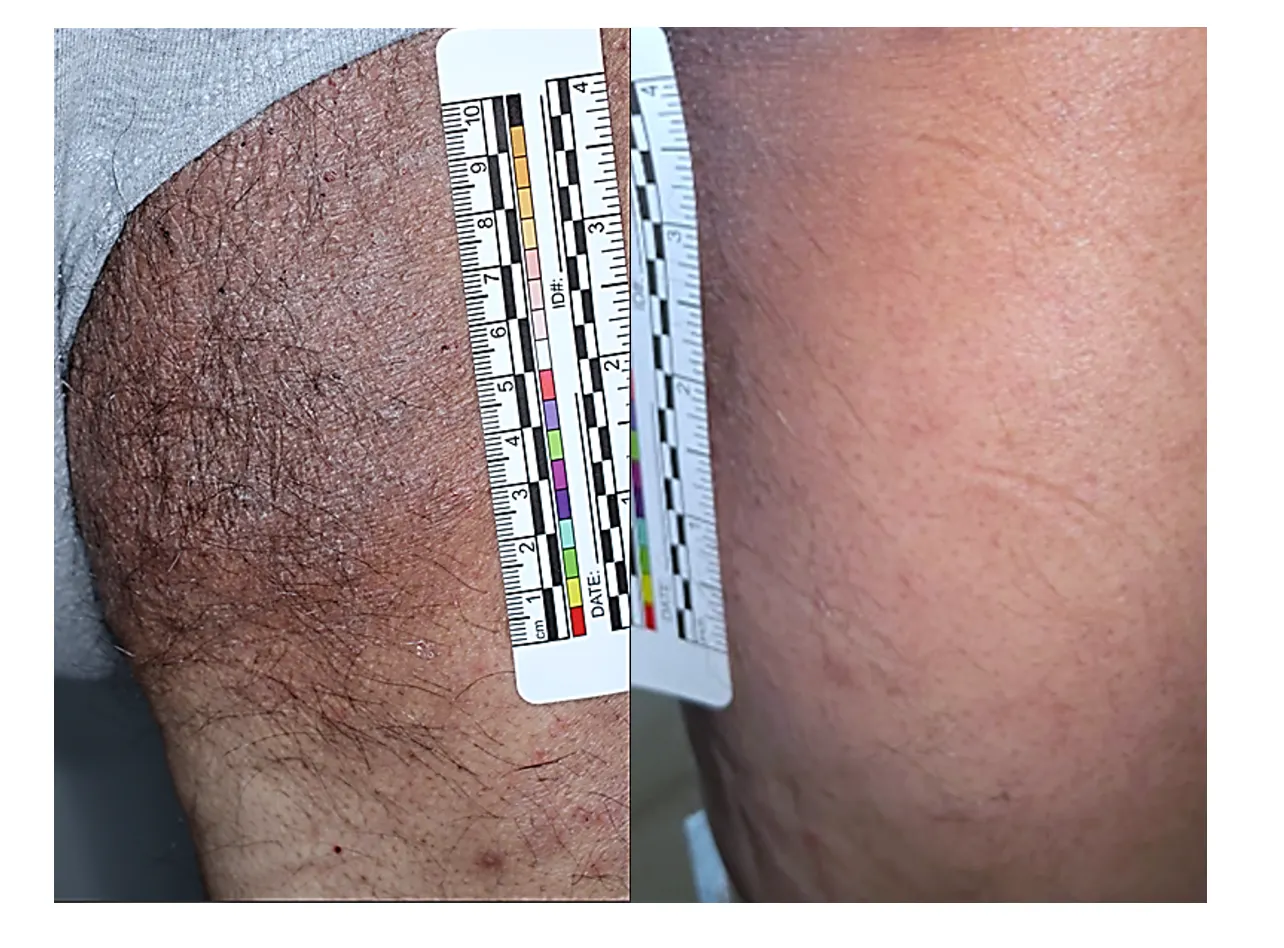
(PASI 90) High responder
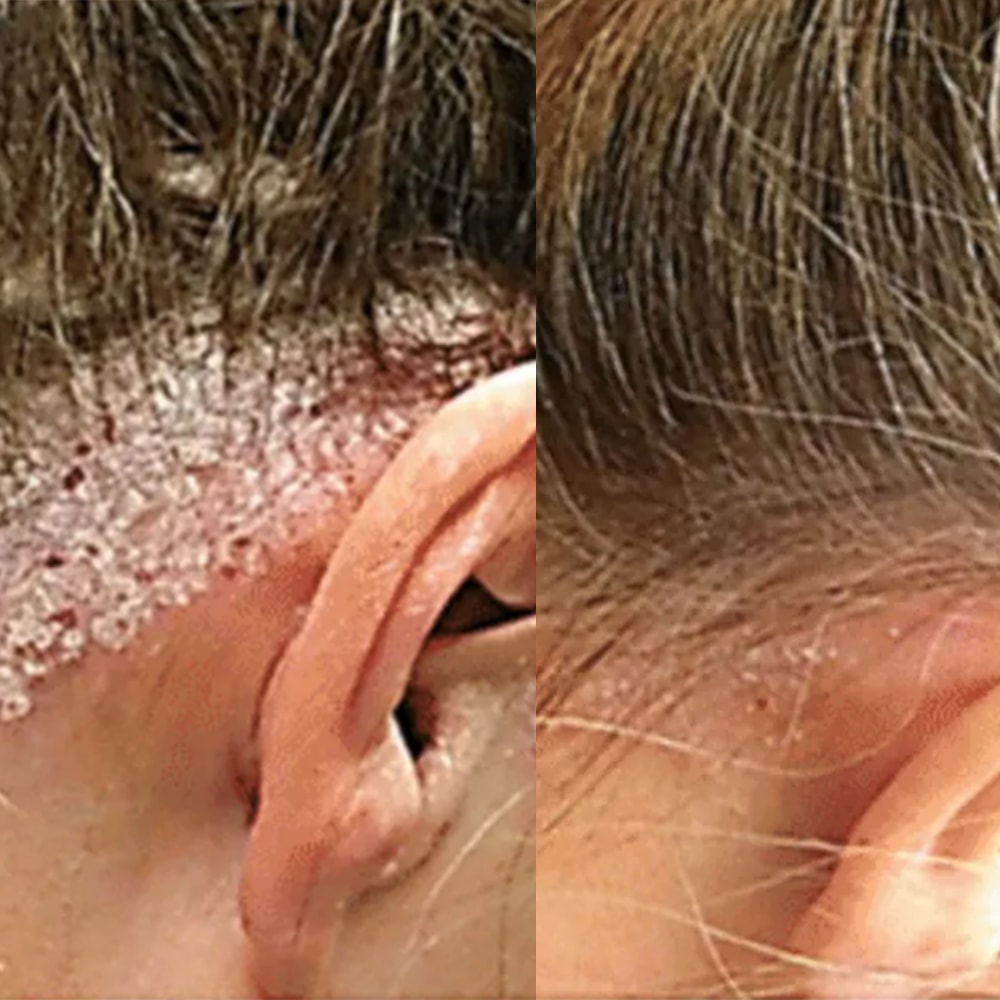
(PASI 75) Typical responder
How It Works
SFA-002 raises interleukin-10 and expands regulatory T cells to support immune tolerance in skin.
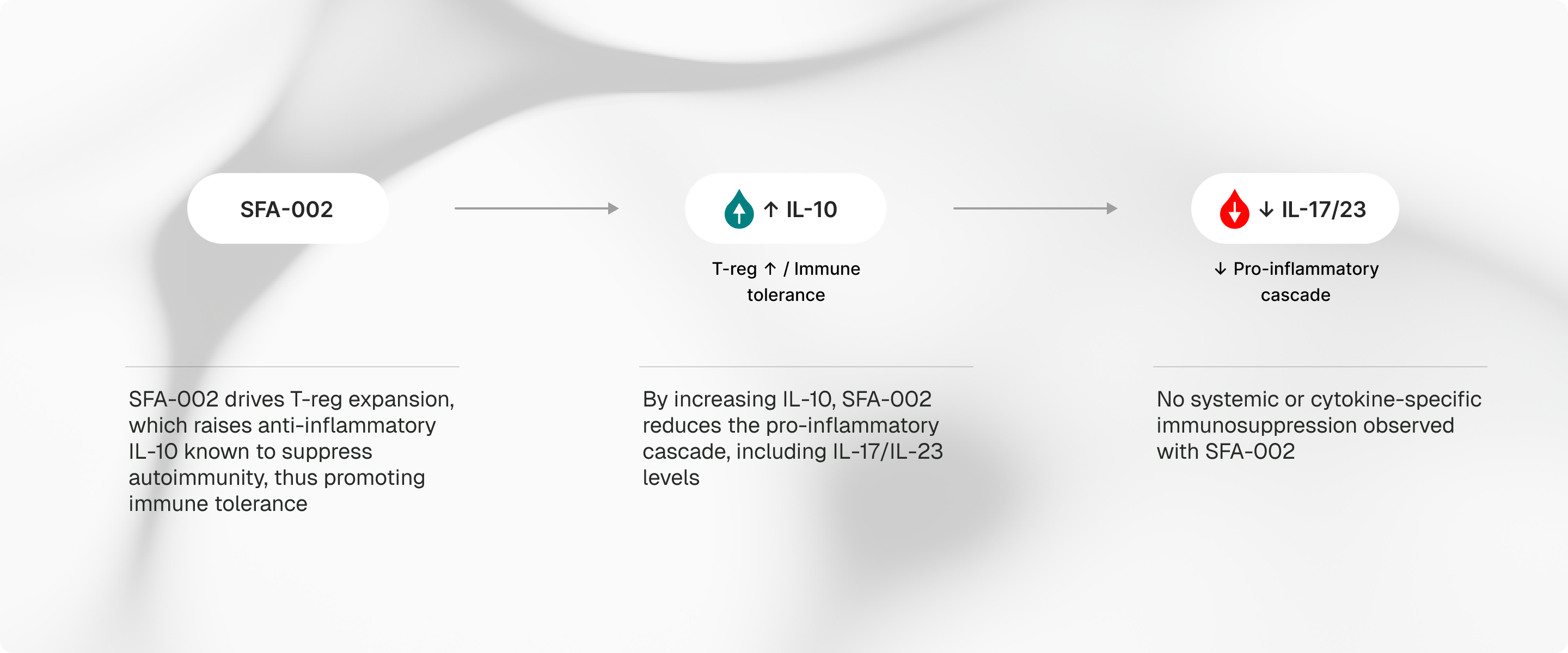
This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Beyond Psoriasis: a platform approach
Based on Nobel Prize-winning research, SFA Therapeutics is developing disease-modifying therapies that restore healthy immune function in patients with autoimmune, inflammatory, and oncologic conditions.
Our patented small molecules are based on naturally occurring microbial metabolites found in healthy people—often reduced in autoimmune conditions. Restoring physiological levels supports durable immune tolerance while preserving normal autoimmune status.Exploring next: Rheumatoid arthritis · Uveitis · Pancreatic cancer adjunct (SFA-009) (all preclinical)
Clinical Trial Status
A randomized, blinded study for mild to moderate psoriasis is in planning. The public registry link will be added when available.
Enrollment
Schedule will be posted when enrollment opens.
Study design
The study is designed to provide a fair measurement of effect size.
Formulation
A deuterated, once-daily tablet is in development, with bridging work planned.
Get study updates >
Clinical Trial Status
Ira C. Spector
Chief Executive Officer; Co‑Founder
Mark Feitelson
PhD, Chief Scientific Officer; Co Founder
Alla Arzumanyan
PhD, Chief Development Officer; Co-Founder
Shawn O'Brien
Chairman

SFA
Therapeutics
We are advancing oral immune therapy for psoriasis.
Work with us
610 Old York Rd, Suite 400, Jenkintown, Pennsylvania 19046, United States
267-584-1080
Restore immune tolerance
2024 by SFA THERAPEUTICS, INC.
For investors

Oral immune tolerance therapy for psoriasis
An investigational oral therapy designed to restore immune tolerance in psoriasis, supported by early human results.
See psoriasis results
Phase 1b Study of SFA-002 | 28 Participants | 12 Weeks
Early human results showed meaningful skin clearance in people with mild to severe plaque psoriasis.
Efficiency
Phase 1B results demonstrated statistically significant response rates competitive with approved oral and biological therapies.
Safety
No treatment-emergent adverse events were observed in 28 participants during the phase 1b, suggesting SFA-002 is highly suitable for mild plaque psoriasis patients.
* Phase 1b, open-label study. Safety assessed through treatment-emergent adverse events.
Individual Results
Results from Phase 1b. Individual results vary.
(PASI 100) Complete clearance

(PASI 90) High responder

(PASI 75) Typical responder

How It Works
SFA-002 raises interleukin-10 and expands regulatory T cells to support immune tolerance in skin.

This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Beyond Psoriasis: a platform approach
Based on Nobel Prize-winning research, SFA Therapeutics is developing disease-modifying therapies that restore healthy immune function in patients with autoimmune, inflammatory, and oncologic conditions.
Our patented small molecules are based on naturally occurring microbial metabolites found in healthy people—often reduced in autoimmune conditions. Restoring physiological levels supports durable immune tolerance while preserving normal autoimmune status.Exploring next: Rheumatoid arthritis · Uveitis · Pancreatic cancer adjunct (SFA-009) (all preclinical)
Clinical Trial Status
A randomized, blinded study for mild to moderate psoriasis is in planning. The public registry link will be added when available.
Enrollment
Schedule will be posted when enrollment opens.
Study design
The study is designed to provide a fair measurement of effect size.
Formulation
A deuterated, once-daily tablet is in development, with bridging work planned.
Get study updates >
Meet the team
Ira C. Spector
Chief Executive Officer; Co‑Founder
Mark Feitelson
PhD, Chief Scientific Officer; Co Founder
Alla Arzumanyan
PhD, Chief Development Officer; Co-Founder
Shawn O'Brien
Chairman

SFA
Therapeutics
We are advancing oral immune therapy for for psoriasis.
Work with us
610 Old York Rd, Suite 400, Jenkintown, Pennsylvania 19046, United States
267-584-1080
Restore immune tolerance
2024 by SFA THERAPEUTICS, INC.
Science
Pipeline
Clinical Trials
Team
Contact
For investors

Oral immune tolerance therapy for psoriasis
An investigational oral therapy designed to restore immune tolerance in psoriasis, supported by early human results.
See psoriasis results
Phase 1b Study of SFA-002 | 28 Participants | 12 Weeks
Early human results showed meaningful skin clearance in people with mild to severe plaque psoriasis.
Efficiency
Phase 1B results demonstrated statistically significant response rates competitive with approved oral and biological therapies.
Safety
No treatment-emergent adverse events were observed in 28 participants during the phase 1b, suggesting SFA-002 is highly suitable for mild plaque psoriasis patients.
* Phase 1b, open-label study. Safety assessed through treatment-emergent adverse events.
Individual Results
Results from Phase 1b. Individual results vary.
(PASI 100) Complete clearance

(PASI 90) High responder

(PASI 75) Typical responder

How It Works
SFA-002 raises interleukin-10 and expands regulatory T cells to support immune tolerance in skin.

This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Beyond Psoriasis: a platform approach
Based on Nobel Prize-winning research, SFA Therapeutics is developing disease-modifying therapies that restore healthy immune function in patients with autoimmune, inflammatory, and oncologic conditions.
Our patented small molecules are based on naturally occurring microbial metabolites found in healthy people—often reduced in autoimmune conditions. Restoring physiological levels supports durable immune tolerance while preserving normal autoimmune status.Exploring next: Rheumatoid arthritis · Uveitis · Pancreatic cancer adjunct (SFA-009) (all preclinical)
Clinical Trial Status
A randomized, blinded study for mild to moderate psoriasis is in planning. The public registry link will be added when available.
Enrollment
Schedule will be posted when enrollment opens.
Study design
The study is designed to provide a fair measurement of effect size.
Formulation
A deuterated, once-daily tablet is in development, with bridging work planned.
Get study updates >
Meet the team
Ira C. Spector
Chief Executive Officer; Co‑Founder
Mark Feitelson
PhD, Chief Scientific Officer; Co Founder
Alla Arzumanyan
PhD, Chief Development Officer; Co-Founder
Shawn O'Brien
Chairman

SFA
Therapeutics
We are advancing oral immune therapy for psoriasis.
Work with us
610 Old York Rd, Suite 400, Jenkintown, Pennsylvania 19046, United States
267-584-1080
Restore immune tolerance
2024 by SFA THERAPEUTICS, INC.
Science
Pipeline
Clinical Trials
Team
Contact
For investors

Oral immune tolerance therapy for psoriasis
An investigational oral therapy designed to restore immune tolerance in psoriasis, supported by early human results.
See psoriasis results
Phase 1b Study of SFA-002 | 28 Participants | 12 Weeks
Early human results showed meaningful skin clearance in people with mild to severe plaque psoriasis.
Efficiency
Phase 1B results demonstrated statistically significant response rates competitive with approved oral and biological therapies.
Safety
No treatment-emergent adverse events were observed in 28 participants during the phase 1b, suggesting SFA-002 is highly suitable for mild plaque psoriasis patients.
* Phase 1b, open-label study. Safety assessed through treatment-emergent adverse events.
Individual Results
Results from Phase 1b. Individual results vary.
(PASI 100) Complete clearance

(PASI 90) High responder

(PASI 75) Typical responder

How It Works
SFA-002 raises interleukin-10 and expands regulatory T cells to support immune tolerance in skin.

This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Beyond Psoriasis: a platform approach
Based on Nobel Prize-winning research, SFA Therapeutics is developing disease-modifying therapies that restore healthy immune function in patients with autoimmune, inflammatory, and oncologic conditions.
Our patented small molecules are based on naturally occurring microbial metabolites found in healthy people—often reduced in autoimmune conditions. Restoring physiological levels supports durable immune tolerance while preserving normal autoimmune status.Exploring next: Rheumatoid arthritis · Uveitis · Pancreatic cancer adjunct (SFA-009) (all preclinical)
Clinical Trial Status
A randomized, blinded study for mild to moderate psoriasis is in planning. The public registry link will be added when available.
Enrollment
Schedule will be posted when enrollment opens.
Study design
The study is designed to provide a fair measurement of effect size.
Formulation
A deuterated, once-daily tablet is in development, with bridging work planned.
Get study updates >
Meet the team
Ira C. Spector
Chief Executive Officer; Co‑Founder
Mark Feitelson
PhD, Chief Scientific Officer; Co Founder
Alla Arzumanyan
PhD, Chief Development Officer; Co-Founder
Shawn O'Brien
Chairman

SFA
Therapeutics
We are advancing oral immune therapy for psoriasis.
We are advancing oral immune therapy for for psoriasis.
Work with us
610 Old York Rd, Suite 400, Jenkintown, Pennsylvania 19046, United States
267-584-1080
Restore immune tolerance
2024 by SFA THERAPEUTICS, INC.
Science
Pipeline
Clinical Trials
Team
Contact
For investors

Oral immune tolerance therapy for psoriasis
An investigational oral therapy designed to restore immune tolerance in psoriasis, supported by early human results.
See psoriasis results
Phase 1b Study of SFA-002 | 28 Participants | 12 Weeks
Early human results showed meaningful skin clearance in people with mild to severe plaque psoriasis.
Efficiency
Phase 1B results demonstrated statistically significant response rates competitive with approved oral and biological therapies.
Safety
No treatment-emergent adverse events were observed in 28 participants during the phase 1b, suggesting SFA-002 is highly suitable for mild plaque psoriasis patients.
* Phase 1b, open-label study. Safety assessed through treatment-emergent adverse events.
Individual Results
Results from Phase 1b. Individual results vary.
(PASI 100) Complete clearance

(PASI 90) High responder

(PASI 75) Typical responder

How It Works
SFA-002 raises interleukin-10 and expands regulatory T cells to support immune tolerance in skin.

This is associated with reductions in interleukin-17, interleukin-23, interferon-gamma, and tumor necrosis factor, while aiming to avoid broad immunosuppression.
Beyond Psoriasis: a platform approach
Based on Nobel Prize-winning research, SFA Therapeutics is developing disease-modifying therapies that restore healthy immune function in patients with autoimmune, inflammatory, and oncologic conditions.
Our patented small molecules are based on naturally occurring microbial metabolites found in healthy people—often reduced in autoimmune conditions. Restoring physiological levels supports durable immune tolerance while preserving normal autoimmune status.Exploring next: Rheumatoid arthritis · Uveitis · Pancreatic cancer adjunct (SFA-009) (all preclinical)
Clinical Trial Status
A randomized, blinded study for mild to moderate psoriasis is in planning. The public registry link will be added when available.
Enrollment
Schedule will be posted when enrollment opens.
Study design
The study is designed to provide a fair measurement of effect size.
Formulation
A deuterated, once-daily tablet is in development, with bridging work planned.
Get study updates >
Meet the team
Ira C. Spector
Chief Executive Officer; Co‑Founder
Mark Feitelson
PhD, Chief Scientific Officer; Co Founder
Alla Arzumanyan
PhD, Chief Development Officer; Co-Founder
Shawn O'Brien
Chairman

SFA
Therapeutics
We are advancing oral immune therapy for for psoriasis.
Work with us
610 Old York Rd, Suite 400, Jenkintown, Pennsylvania 19046, United States
267-584-1080
Restore immune tolerance
2024 by SFA THERAPEUTICS, INC.




